Investment summary: Tcelna trials ongoing for SPMS
Company description: Calming T-cells with self-proteins
Established in 2004, Opexa is focused on applying the proprietary ImmPath immunotherapy platform in-licensed from Baylor College of Medicine, to poorly served autoimmune diseases. ImmPath is a personalized immunotherapy using a patient’s own blood samples to help down-regulate the dominant T-cells specific to that individual, which are responsible for or highly contributory to disease processes. Lead product Tcelna is in Phase IIb studies for secondary progressive multiple sclerosis (SPMS), with data anticipated in H216. OPX-212 is a separate product candidate and in preclinical development for neuromyelitis optica (NMO). Merck KGaA has an option to in-license Tcelna by the end of the Phase IIb program.
Exhibit 1: Opexa Therapeutics upcoming catalysts
Event |
Timing |
IND filing for OPX-212 Phase I/II NMO study |
H116 |
Start OPX-212 Phase I/II study |
Mid-2016 |
Results from Abili-T Phase IIb SPMS study |
H216 |
Event |
IND filing for OPX-212 Phase I/II NMO study |
Start OPX-212 Phase I/II study |
Results from Abili-T Phase IIb SPMS study |
Timing |
H116 |
Mid-2016 |
H216 |
Source: Edison Investment Research, company reports
Valuation: Equity value of $51m presents significant upside
Our rNPV-derived equity valuation of $51m (inclusive of $15.6m net cash at Q315), or $7.39 fully diluted, applies a 12.5% cost of capital and assumes a 15% probability of success for Tcelna in SPMS and a 5% probability for OPX-212 in NMO. We assume global Tcelna peak sales by partner Merck KGaA of $2.6bn in 2026, with Opexa entitled to 8-15% in tiered royalties.
Financials: Funded through Phase IIb SPMS data
On 30 September 2015, Opexa had $15.6m in cash and equivalents and its trailing 18-month cash burn rate was $17.3m. In September 2015, the firm raised $0.5m from a $5m equity facility dedicated to OPX-212 (we expect the remaining $4.5m to be raised by year end 2017). We project quarterly burn rates of $3.2-3.7m in the coming quarters as the firm completes the Tcelna Phase IIb study and advances OPX-212. If Tcelna Phase IIb data, expected in H216, are positive, Merck KGaA can exercise its option to in-license Tcelna, which would trigger a $25m upfront payment to Opexa. Merck KGaA would then fund the remainder of Tcelna studies in MS. We forecast that Opexa will raise $60m between 2018 and 2020 to advance OPX-212 and potentially other ImmPath programs. This assumes Merck KGaA exercises its licence option; otherwise, we expect Opexa will need to raise an additional $5m in Q416 and $10m in 2017. For illustrative purposes, we assign these fund-raisings to long-term debt.
Sensitivities: Funding, development risks, competition
While early pooled open-label Phase I SPMS data show a slower than average rate of disease progression, the condition is notoriously difficult to treat, with numerous clinical trial failures (including the recently reported natalizumab Phase III SPMS study) and only one treatment being approved by the FDA. Altogether, there remains significant development and regulatory risk for emerging treatments in neurodegenerative conditions such as MS or NMO. An additional challenge will be sustaining access to capital at favourable terms to fund OPX-212 and future ImmPath programs through multiple clinical trials, and/or to further advance Tcelna if Merck KGaA does not exercise its option. Competing products are also being developed for SPMS and could potentially reach the market earlier than Tcelna, and Tcelna’s commercial success will depend on its performance vs potential competing new products.
Multiple sclerosis overview
MS is an inflammatory disease that attacks the central nervous system. It erodes the protective myelin sheath around nerve fibres, impairing neural signal conduction, and may also attack the nerve fibres themselves. Patients with MS experience a range of neurological dysfunction such as paralysis, ataxia, vision and other sensory losses, in addition to dementia and psychological symptoms. According to the Cleveland Clinic, MS affects approximately 400,000 people in the US and 2.5 million worldwide. Onset is usually between 20 and 40 years and the disease tends to affect women twice as frequently as men. MS is most frequently described in one of three forms:
■
relapsing-remitting MS (RRMS) is the most common form of the disease, accounting for 85-90% of the initial diagnosis. In RRMS, neurologic symptoms appear periodically for several days to weeks (relapse), after which they usually resolve spontaneously (remittance).
■
primary progressive MS (PPMS) affects approximately 10-15% of patients. These patients have gradually worsening symptoms from the onset without clinical remissions.
■
secondary progressive MS (SPMS) is considered a later-stage form of RRMS. SPMS is marked by progressive neurologic decline, similar to PPMS. According to the National Multiple Sclerosis Society, historically 50% of people diagnosed with RRMS develop SPMS within 10 years, and 90% transition within 25 years.
A fourth form, progressive relapsing MS, is considered to be a variant of SPMS, where the initial relapses/remittance were very subtle or asymptomatic.
Exhibit 2: Different forms of multiple sclerosis by clinical characteristics
|
Exhibit 3: Transition between RRMS and SPMS
|

|
|
Source: Adapted from Lublin FD, et al. Defining the clinical course of multiple sclerosis: results of an international survey. Neurology 1996; 46:907–911.
|
Source: Biogen Investor Presentation, 3 November 2015
|
Exhibit 2: Different forms of multiple sclerosis by clinical characteristics
|

|
Source: Adapted from Lublin FD, et al. Defining the clinical course of multiple sclerosis: results of an international survey. Neurology 1996; 46:907–911.
|
Exhibit 3: Transition between RRMS and SPMS
|

|
Source: Biogen Investor Presentation, 3 November 2015
|
MS is believed to be an autoimmune disorder, as immune cells targeting myelin components and associated inflammation factors or mediators (cytokines and antibodies) are involved. Most existing approved disease-modifying treatments for MS (including interferon-beta, glatiramer acetate and natalizumab) aim to modulate immune system function to reduce attacks against myelin and nearly all these are specifically approved for RRMS only.
Tcelna intends to impede the inflammatory processes of MS by targeting and suppressing myelin-reactive T-cells (MRTCs) believed to be responsible in modulating inflammatory processes. After blood is extracted from an MS patient, PBMCs are isolated and screened for T-cells (MRTCs) against a proprietary bank of 109 different myelin peptides. These include peptides from myelin basic protein (MBP), myelin oligodendrocyte glycoprotein (MOG) and proteolipid protein (PLP). The process then identifies which specific peptide antigens are being targeted by the patient’s own MRTCs. Management estimates that out of 109 peptides screened by the process, typically MRTCs against three or four peptides are dominant.
An autologous pool of MRTCs is then generated and raised against these selected peptides. The treatment principle is that the dosage of these MRTCs (after being attenuated with gamma radiation) initiate an immune response that depletes and/or suppresses circulating MRTCs that carry similar peptide-specific T-cell receptor molecules. Hence, Tcelna attempts to impede the function of specific subsets of personalized MRTCs believed to attack myelin and related components.
The Tcelna approach screens patients’ blood each year (against the 109 myelin peptides), as the myelin peptide antigens targeted by the patient’s own endogenous MRTCs can change over time. Effectively, the patient’s own epitope profile is re-analysed at each year of therapy, thus re-customizing the treatment to account for potential “epitope drift”.
Previous studies show evidence of MRTC reductions
A 16-patient, Phase I/II open-label study in MS patients intolerant or unresponsive to existing treatments showed statistically significant MRTC cell count reductions over baseline at all visits through one year (five doses given per year, at weeks 0, 5, 13, 21 and 28). Tcelna doses were comprised of attenuated MRTC cells selected against six myelin peptides (two each from MBP, PLP and MOG). Three dose levels were tested (6-9m cells per dose, 30-45m cells per dose, 60-90m cells per dose), and the effect was strongest in the middle dose range. There were no reports that MRTC changes for RRMS patients (n=9) differed from those of SPMS patients (n=7).
Exhibit 4: Change in MRTCs (%) in blood from baseline of patients by dose in evaluable per-protocol population (Phase I/II study)
|
Week 5 |
Week 13 |
Week 21 |
Week 28 |
Week 52 |
6-9m cells/dose cohort; n=6 |
-46.4 |
-56 |
-30.4 |
-67.8 |
-35.4 |
30-45m cells/dose cohort; n=5 |
-92.4 |
-86.9 |
-79.4 |
-76.7 |
-64.8 |
60-90m cells/dose cohort; n=5 |
-77.3 |
72.3 |
-54.1 |
-22.8 |
18.3 |
Source: Clin Immunol. 2009 May;131(2):202-15
In the high-dose group, the data were skewed by a high MRTC count increase for one subject. This could suggest a dose-limiting effect. Opexa used 30-45m cells per dose for subsequent studies. The data indicate that Tcelna causes immediate depletion of these MRTCs, which subsides in the weeks following dosing. A subset of the above patients was followed in an extension study, and of these, those who had a rebound of MRTC were given a subsequent annual Tcelna course and then showed continued significant reductions over baseline in the MRTC counts.
TERMS Phase IIb RRMS missed primary endpoint but showed other signals
A 150-patient, 12-month, placebo-controlled Phase IIb study of Tcelna in RRMS (Tovaxin for Early Relapsing Multiple Sclerosis, TERMS) in RRMS or Clinically Isolated Syndrome was completed in 2008. Patients received five subcutaneous Tcelna injections (at weeks 0, 4, 8, 12 and 24).This study did not show statistical significance in its primary endpoint, cumulative number of gadolinium-enhanced brain lesions using magnetic resonance imaging (MRI). However, in a modified intent-to-treat patient population consisting of patients who received at least one dose of study product and had at least one MRI scan at week 28 or later (n=142), the annualized relapse rate (ARR), a measure of RRMS progression, for Tcelna-treated patients was 0.214 compared to 0.339 for placebo-treated patients, representing a 37% decrease in ARR for Tcelna vs placebo. This decrease in relapse rates vs placebo is comparable to those of approved RRMS drugs.
Exhibit 5: Comparison of effect on relapses between TERMS study subset (n=142) and selected approved RRMS therapies
Product |
Reduction in relapses vs placebo |
Time period |
Source |
Tcelna (n=142) |
37% |
12 months |
Opexa reports |
Interferon Beta-1b (n=372) |
34% |
24 months |
FDA Betaseron prescribing information |
Interferon Beta-1a (n=301) |
29% |
24 months |
Jacobs LD, Cookfair DL, Rudick RA, et al. Ann Neurol. 1996 Mar. 39(3):285-94 |
Peginterferon beta-1a (n=1512) |
36% |
12 months |
Calabresi PA, Kieseier BC, Arnold DL, et al. Lancet Neurol. 2014 Jul. 13(7):657-65 |
Glatiramer acetate (n=251) |
29% |
24 months |
Johnson KP, Brooks BR, Cohen JA, et al. Neurology. 1995 Jul. 45(7):1268-76 |
Natalizumab (n=942) |
68% |
12 months |
Polman CH, O'Connor PW, Havrdova E et al. N Engl J Med. 2006 Mar 2. 354(9):899-910 |
Dimethyl fumarate (n=1234) |
53% |
24 months |
Gold R, Kappos L, Arnold DL, et al. N Engl J Med. 2012 Sep 20. 367(12):1098-107 |
Source: Edison Investment Research, Medscape. Note: Excluding Tcelna, this table compiles data from the placebo-controlled pivotal Phase III studies used in regulatory applications for the listed RRMS drugs.
In a prospective group of patients (n=50) with more active disease at baseline (ARR>1), the Tcelna-treated group demonstrated a 55% reduction in ARR vs placebo, and a significant improvement in disability (using Expanded Disability Status Scale, EDSS) vs placebo (p<0.045). A pooled analysis from 36 SPMS patients from earlier Phase I open-label studies (two Opexa-sponsored studies completed in 2006 and 2007 and one by Baylor in 1998) showed that c 80% of those completing two years of Tcelna showed no disease progression as measured through EDSS. Historical control data (ESIMS study, published by Hommes et al. in Lancet in 2004) suggest that 40% of SPMS patients would normally progress. This, combined with the stronger efficacy signal in patients with more active RRMS in TERMS, helped lead the company to pursue SPMS for future Tcelna studies.
Pathophysiology rationales for Tcelna in SPMS
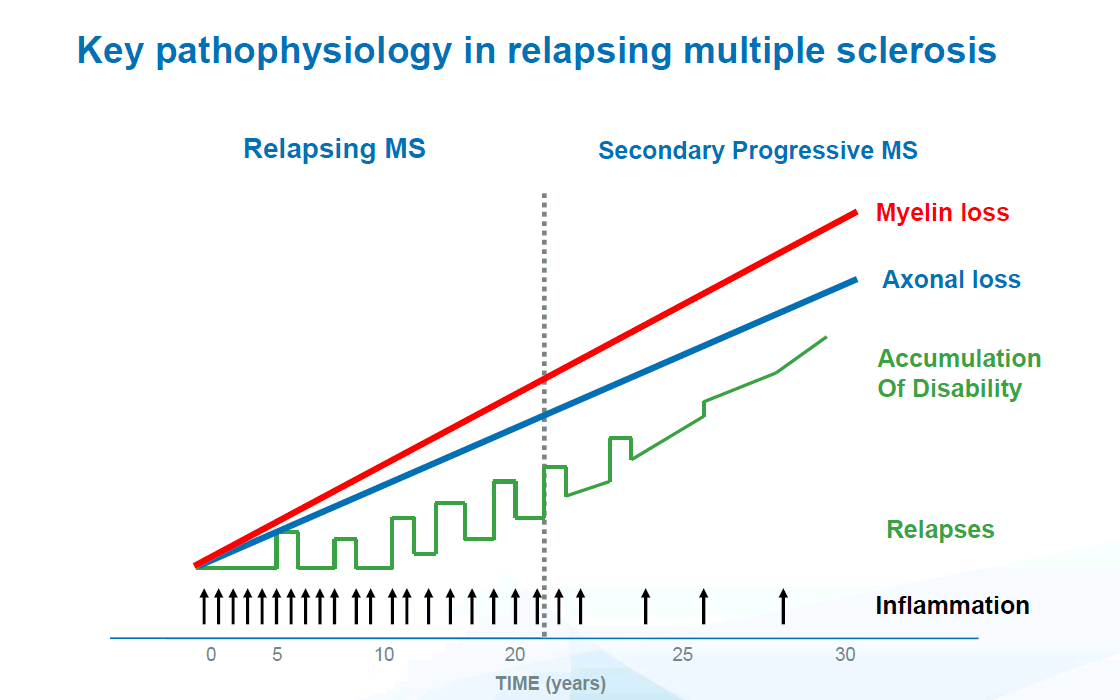
Although SPMS develops from RRMS, it exhibits many PPMS characteristics. Where RRMS is characterised by active inflammation, an increase in lesions and evidence of remyelination, SPMS is characterised by more diffuse and less intense inflammation, little to no expansion in the number or size of lesions, and sparse evidence of remyelination. SPMS patents tend to have significantly more gray matter atrophy compared with RRMS patients.
The reduced level of acute inflammation could help explain how available immunomodulating and immunosuppressive therapies approved and validated for RRMS have not been effective in SPMS.
Exhibit 6: Pathophysiological differences between RRMS and SPMS/PPMS
RRMS |
SPMS/RRMS |
Active lesions |
Chronic lesions |
Active expansion of new and existing lesions |
Slow expansion of pre-existing lesions |
Axonal damage focused in demyelinating lesions |
Diffuse axonal damage (not concentrated in lesions) |
Lesions are diffuse in white matter of brain |
Grey matter of brain more affected |
Evidence of remyelination of certain lesions |
Inflammation behind BBB |
|
Meningeal inflammatory aggregates |
|
Brain atrophy |
RRMS |
Active lesions |
Active expansion of new and existing lesions |
Axonal damage focused in demyelinating lesions |
Lesions are diffuse in white matter of brain |
Evidence of remyelination of certain lesions |
|
|
SPMS/RRMS |
Chronic lesions |
Slow expansion of pre-existing lesions |
Diffuse axonal damage (not concentrated in lesions) |
Grey matter of brain more affected |
Inflammation behind BBB |
Meningeal inflammatory aggregates |
Brain atrophy |
Source: Edison Investment Research, Lassmann, H. et al. Nat. Rev. Neurol. 8, 647-656 (2012); published online 25 September 2012; doi:10.1038/nrneurol.2012.168
SPMS pathology may involve both the adaptive and innate immune system (whereas RRMS pathology is driven by adaptive immune cells). Unlike RRMS, cells of the innate immune system, including persistently activated microglia and dendritic cells, are dominant in progressive MS, and lymphoid follicles in the meninges (close to gray matter) are also present. Opexa believes that the transition to SPMS may involve a transition to a T-cell-dependent inflammatory mechanism, which may more heavily involve the MRTCs as targeted by Tcelna. This could potentially provide a rationale for Tcelna to provide more efficacy in SPMS than RRMS.
Opexa believes that Tcelna’s down-regulatory responses against the overrepresented MRTCs can in turn down-regulate similar (but not identical) alternate endogenous disease-causing MRTCs. It believes that Tcelna up-regulates Foxp3 (Forkhead Box P3) and Interleukin-10 secreting regulatory T-cells, both of which have been shown in animal studies to improve self-tolerance (and reduce autoimmunity). The company cites that among TERMS patients who had baseline ARR>1, Tcelna-treated patients had statistically significant changes from baseline (p=0.02) in Foxp3 cells.
Blood-brain barrier issue an alternative explanation for SPMS difficulties
One complicating factor for SPMS, which may also explain why existing RRMS-approved drugs are unsuccessful, is that the blood-brain barrier (BBB) is less disrupted (and more impermeable) in SPMS compared to RRMS, which may impede the ability of therapeutics to reach target sites; indeed, the (chronic) inflammation and inflammatory cells surrounding SPMS may effectively be “trapped behind the BBB” and less reachable by proposed experimental treatments.
Abili-T Trial: Phase IIb clinical study in patients with SPMS
In fall 2012, Opexa initiated a 190-patient, placebo-controlled (1:1 randomization) Phase IIb study (termed Abili-T) of Tcelna in patients with SPMS and recruitment was completed in June 2014. The trial is being conducted at 35 US and Canadian sites and recruited patients must have EDSS scores between 3.0 and 6.0, evidence of ongoing SPMS progression and measurable MRTCs in their bloodstream. Patients receive two annual courses of Tcelna treatment consisting of five subcutaneous injections per year at weeks 0, 4, 8, 12 and 24. The primary endpoint is the percentage of brain volume change (whole brain atrophy) at 24 months, and EDSS change and ARR are included as secondary endpoints. The firm will also monitor pro-inflammatory and anti-inflammatory immune biomarkers for patients in the study. The firm expects top-line data in H216. We have limited information as to whether Tcelna has shown effects on atrophy in SPMS patients treated in prior studies. If Abili-T data are positive and Tcelna proceeds to pivotal Phase III SPMS trials, we anticipate that the primary endpoint would be EDSS change.
Merck KGaA option to in-license Tcelna
In February 2013, Merck KGaA paid $5m upfront to Opexa to acquire an option to obtain an exclusive worldwide licence (excluding Japan) to the Tcelna program for the treatment of MS. Merck KGaA has existing familiarity with the MS drug development and commercialisation, as it markets Rebif (interferon beta-1a) for RRMS.
Merck KGaA can exercise this option before or after the completion of the ongoing SPMS Abili-T study. If the option is exercised, Merck would pay a $25m upfront licence fee, unless additional Phase II studies are required (as determined by Merck KGaA) to bring Tcelna to Phase III studies, which would reduce the upfront fee to $15m. Following the exercise, Opexa would be entitled to tiered royalties on Tcelna commercialisation, at rates in a range of 8-15% of net sales, with step-ups occurring in this range when annual net sales exceed $0.5bn, $1.0bn and $2bn.
Merck KGaA would also be responsible for funding all future development, regulatory and commercial activities, as well as product manufacturing costs. Opexa would be entitled to up to $195m of additional milestones, with up to $70m achievable for regulatory approvals or launches in SPMS ($35m for US, up to $30m for Europe and $5m for other regions), up to $40m for approvals or launches in RRMS and up to $85m in commercial milestones (with $55m achievable on annual net sales exceeding $1bn).
Competition analysis in SPMS
Current RRMS drugs are not approved for use in SPMS, and the only FDA US-approved SPMS product is mitoxantrone, an immunosuppressive drug that was originally developed and approved as a treatment for certain cancers. Mitoxantrone’s use in SPMS settings is limited by its propensity to cause irreversible cardiomyopathy. Several other products are in the pipeline for the treatment of progressive MS, including SPMS. The commercial success of such candidates could influence Tcelna’s potential market share and reach if it gains approval.
Exhibit 7: Selected NMEs in development of Progressive MS and/or SPMS
Product |
Company |
Mechanism |
Current stage |
Notes |
Clinical
trial ID |
|
|
|
|
|
|
Ocrelizumab |
Roche |
Humanized Anti-CD20 mAb; targets mature CD20-positive B-lymphocytes; CD20-positive B-cells are proposed to strongly contribute to myelin and axonal damage in MS patients. |
Phase III |
In 732-pt PPMS Phase III, ocrelizumab significantly reduced progression of clinical disability (EDSS) sustained for at least 12 weeks (primary endpoint) and 24 weeks vs placebo. Regulatory submissions for RRMS and SPMS are planned for H116. |
NCT01194570 |
BG00012
(Tecfidera) |
Biogen |
Oral dimethyl fumarate that activates the NF-E2-related factor 2 (Nrf2) pathway. |
Phase III |
Investigate whether treatment with BG00012 (dimethyl fumarate) vs placebo slows the accumulation of disability not related to relapses in participants with SPMS. Estimated completion June 2019. |
NCT02430532 |
BAF312
(siponimod) |
Novartis |
Immune-modulating treatment designed to be a more selective sphingosine 1-phosphate (S1P) receptor modulator than Gilenya. Siponimod acts by retaining specific lymphocytes in lymph nodes to prevent them from reaching the central nervous system (CNS). |
Phase III |
Specifically targets receptor subtypes 1 and 5 (S1P1 and S1P5); Only recruiting SPMS patients; Estimated study completion August 2016. |
NCT01665144 |
MD1003 |
Medday |
Concentrated formulation of D-biotin that targets a rate-limiting enzyme in myelination; MD1003 may stimulate myelin production and improve nerve impulse conduction. |
Phase III |
Data expected H116; regulatory applications to FDA and EMA expected in 2016. Reported additional positive data in progressive MS June 2015. |
NCT02220933 |
Masitinib |
AB Science |
Selective tyrosine kinase inhibitor; may affect survival, migration, and degranulation of mast cells by inhibiting key cell signaling pathways; this indirectly controls the amount of pro-inflammatory mediators released in CNS that contribute to MS characteristics. |
Phase IIb/III |
Assessing patients with PPMS or SPMS; study completion near year-end 2015. |
NCT01433497 |
MIS416 |
Innate Immuno-therapeutics |
Immune modulation; MIS416, a microparticle derived from bacteria, may act to stimulate certain responses in the innate immune system. It is believed to suppress pro-inflammatory Th1, Th2 and Th17 immune cells and raise interferon-gamma signalling. |
Phase IIB |
Immunostimulant comprised of caspase recruitment domain family member 15 (CARD15; NOD2) and toll-like receptor 9 (TLR9) ligands. Currently recruiting. Estimated completion June 2016. |
NCT02228213 |
ATX-MS-1467 |
Apitope |
Vaccine containing four synthetic peptides derived from human myelin basic protein. |
Phase IIa |
Open-label baseline proof-of-concept study primarily focused on RRMS, but includes SPMS patients. Completed Phase IIa enrollment September 2015. Data expected 2016. Partnership with Merck Serono. |
NCT01973491 |
IR 1902
(Neurovax) |
Immune Response BioPharma |
Neurovax is designed to stimulate Foxp3+ regulatory T-cells, which can then potentially suppress the autoreactive T-cells in some patients. |
Phase II |
Original development program halted in 2008 when predecessor company filed for bankruptcy. Anticipated study start date March 2016. Anticipated end date March 2018. |
NCT02149706 |
BIIB033 |
Biogen |
anti-LINGO-1 mAb; LINGO-1 may inhibit myelination, neuronal survival, axonal regeneration, and oligodendrocyte differentiation. Anti-LINGO-1 inhibits LINGO-1 in an attempt to promote neuroprotection and remyelination in MS patients. |
Phase II |
LINGO-1 looks at efficacy and tolerability of BIIB033 when used concurrently with Avonex. Patients must have RRMS or onset of SPMS. Estimated study completion June 2016. |
NCT01864148 |
MN166
(ibudilast) |
Kyorin Pharma |
Orally bioavailable, small molecule glial attenuator that suppresses pro-inflammatory cytokines IL-1ß, TNF-a, and IL-6, and may up-regulate the anti-inflammatory cytokine IL-10. |
Phase II |
Phase II interim data expected Q416 (full data H117). Partnership with MediciNova. Currently marketed for stroke and asthma. |
NCT01982492 |
Source: Edison Investment Research, clinicaltrials.gov
Roche’s ocrelizumab, which targets CD20-positive B-cells, could be approved for SPMS before Tcelna, given positive Phase III results in PPMS (n=732) reported in September 2015. Ocrelizumab significantly reduced disability progression as measured by EDSS vs placebo. While these data suggest CD20-positive B-cells could play a strong role in the myelin and axonal nerve damage caused by progressive MS, it does not necessarily suggest that MRTCs (as targeted by Tcelna) would be any less involved. Opexa hypothesizes that the involved B-cells could potentially help present antigen to autoreactive T-cells, facilitating MRTC formation propagation. It suggests ocrelizumab’s efficacy in the PPMS study could be driven by the downstream influence of CD20 B-cell depletion on T-cells, and this could bode well for treatments targeting T-cells (such as Tcelna).
Other potentially promising SPMS competitors include Novartis’s siponimod, a more selective sphingosine 1-phosphate (S1P) modulator than fingolimod and in a 1,500-pt Phase III study, and Medday’s MD10003, which reported positive PPMS data (a 67% lower rate of EDSS progression in the treatment arm at 12 months vs placebo; n=144). Biogen’s Anti-LINGO-1 mAb, currently in a Phase II study, is noteworthy for aiming to regrow myelin, rather than just prevent future damage.
OPX-212 for neuromyelitis optica (NMO)
NMO is a rare autoimmune disorder whereby immune cells and antibodies attack astrocytic and myelin cells in the optic nerves and the spinal cord, and for which there is no approved treatment. NMO patients suffer from optic neuritis (acute ocular/oculomotor pain and vision loss) and transverse myelitis, which can cause weakness and sensory loss in the limbs. The National Multiple Sclerosis Society estimates NMO affects approximately 4,000 people in the US and 250k globally.
NMO attacks the Aquaporin 4 (AQP4) protein found on the surface of astrocytes (supportive neurological cells) leading to demyelination and axonal damage. Opexa proposes that OPX-212 may reduce the number and/or regulate AQP4 reactive T-cells (ARTCs), thus reducing the progression of disability. The firm intends to file an IND for OPX-212 in H116 and then may potentially start a Phase I/II study in Q216. In September 2015, the company entered a five-tranche stock purchase agreement with a private investor to fund this program, including the planned Phase I/II study, for up to $5m, following achievement of predefined milestones. The initial tranche raised $0.5m (in equity priced at $0.55/share) and the subsequent tranches (of up to $4.5m) will be priced based at 90% of prevailing Opexa share market prices.








